Solved A diprotic base (B) has pKb values of 3.08 (pKb1) and

By A Mystery Man Writer
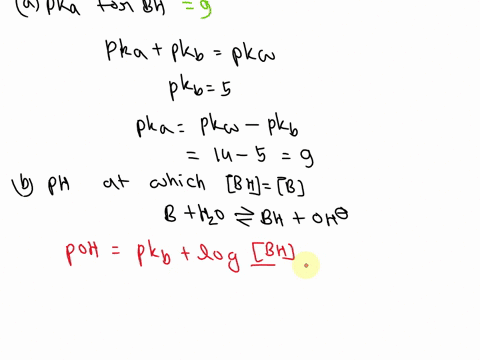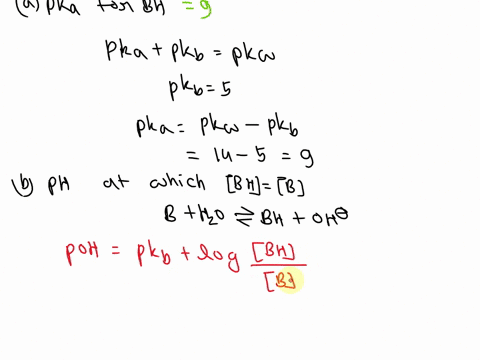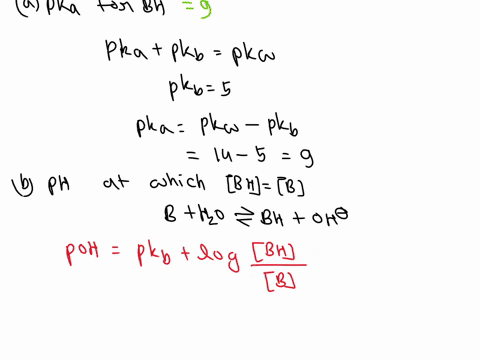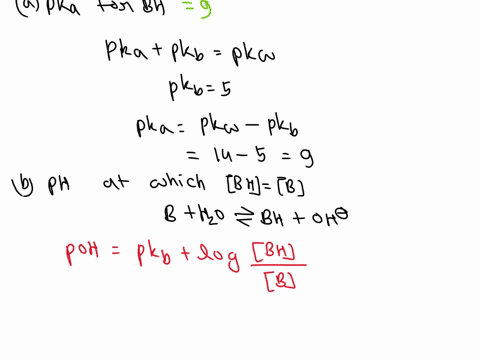
SOLVED: A diprotic base B has pKa values of 3.54, 7.08, and 10.46. Which of the equalities is true when the pH is 6.92? [BH+] = [B] [BH] = [B] [BH+] = [

All 1B CSM PDF, PDF, Reaction Rate
The pKb values for the dibasic base B are pKb1=2.10 and pKb2=7.54. Calculate the pH at each of the points

Diprotic & Polyprotic Acids

Polyprotic Acid-Base Equilibria - ppt video online download

Anachem LEC- Finals notes mls323L short term - CONCEPTS Acids and Bases DEFINITION BY: ACID BASE - Studocu
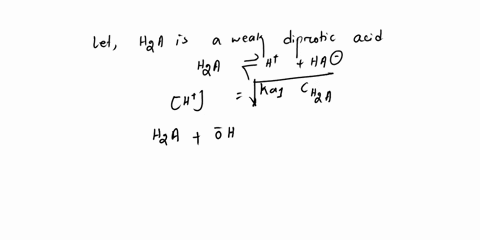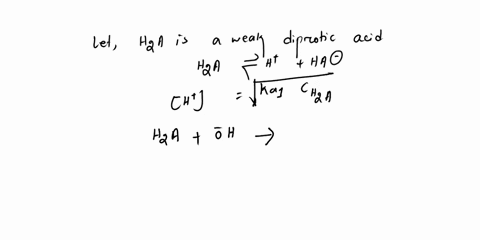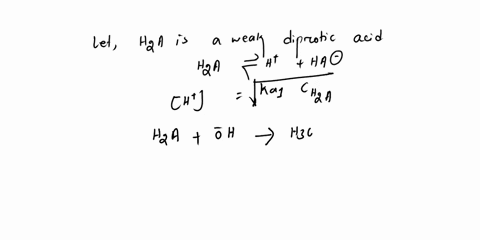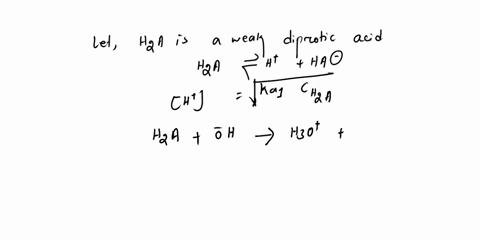
SOLVED: Question 2 of 2: A diprotic base (B) has pKb values of 4.32 and 8.64 (pK62a). Which of the following equalities is true when the pH is 5.36? Which of the

Solved A diprotic base (B) has pKb values of 3.08 (pKb1) and

OneClass: A weak base (B) has a pKb value of 5.77. a) At what pH is [BH ] = [B]? b) What is the predo

OneClass: A weak base (B) has a pKb value of 5.77. a) At what pH is [BH ] = [B]? b) What is the predo

PDF) Quantitative Chemical Analysis

Solved A diprotic base (B) has pKo values of 4.83 (pKb1 and
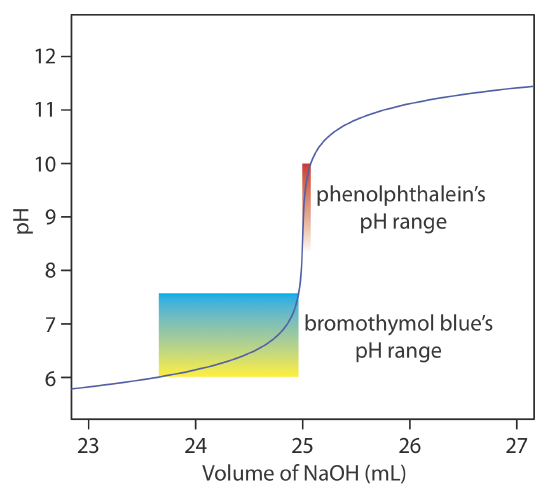
6.6: pH Calculations for Acid–Base Titrations - Chemistry LibreTexts

Solved A diprotic base (B) has pKb values of 3.08 (pKb1) and

OneClass: The pKb values for the dibasic base B are pKb1 = 2.10 and pKb2 =7.85. Calculate the pH at e
- Hilary D, DD, E & F Cup Underwire Bikini Top - Ivory & Black – SKYE

- Bali Women's Leopard Print 2pk T-shirt Bra B202 - Evening Blush 36dd : Target
- alaroo Alaroo Stretch Mermaid Print Fish Scale Leggings Tights

- Lululemon Yellow Jacquard Spacedye Luon Low Rise Wunder Under Crop, Women's 6
- Ladies High Waist Tummy Control Girdle-Tights (BLACK) - Tees Beauty Stores






