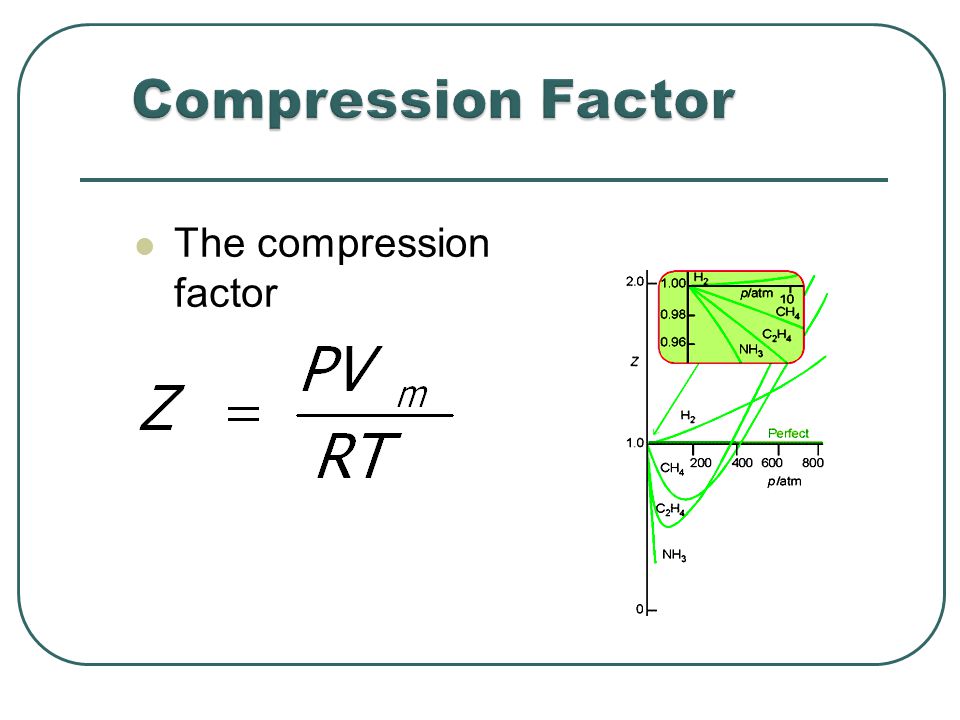
Compression Factor and Fugacity

By A Mystery Man Writer
Share your videos with friends, family and the world
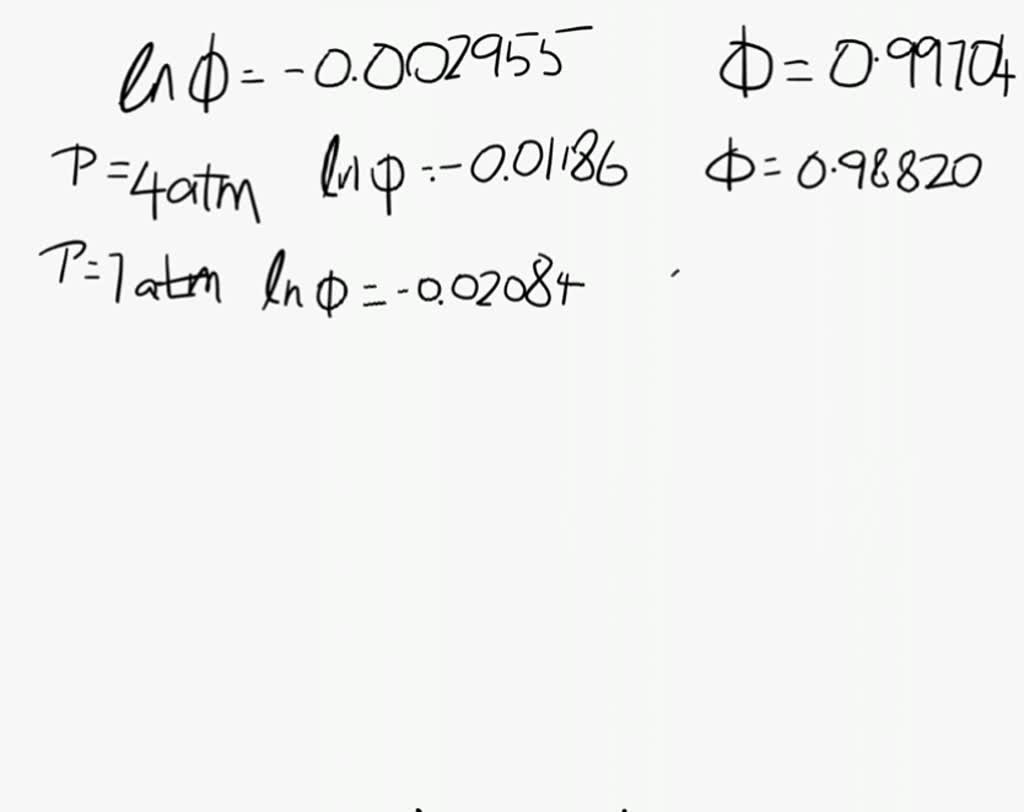
SOLVED: At 200 K, the compression factor of oxygen varies with pressure as shown below. Evaluate the fugacity of oxygen at this temperature and 100 atm: p (atm) Z 1,0000 0.9971 4.00000 0.98796 7,00000 0.97880 10,0000 0.96956 40.00 0.8734 70.00 0.7764

Chemical Thermodynamics 6.10 - Fugacity Coefficient

CHEM 111.1 Ex2 full report.docx - Exercise 2 COMPUTATION OF THE COMPRESSION FACTOR AND FUGACITY OF REAL GASES Ella Mae E. Gamba Kennedy P. Limbo King

Computation of The Compression Factor and Fugacity Coefficient of Real Gases, PDF, Gases

PDF) Z Factor: Implicit Correlation, Convergence Problem and Pseudo-Reduced Compressibility

PPT - Chemistry 231 PowerPoint Presentation, free download - ID

Determination of compressibility factor and fugacity coefficient of hydrogen in studies of adsorptive storage - ScienceDirect
If you increase the pressure of a gas, does its fugacity coefficient increase, decrease, or remain the same? - Quora

Computation of The Compression Factor and Fugacity Coefficient of Real Gases, PDF, Gases